When just one abnormal plasma cell in the bone marrow – the soft, blood-producing tissue that fills in the center of most of the bones – suddenly begins to multiply rapidly, multiple myeloma results.
This is a dangerous cancer that forms in a type of white blood cell that, when healthy, help you fight infections by making antibodies to recognize and attack germs.
Multiple myeloma causes cancer cells to accumulate in the bone marrow, where they crowd out healthy blood cells. Instead of producing antibodies, the cancer cells manufacture abnormal proteins that can cause complications. Doctors know this, but they don’t know why the plasma cells go wild.
If the patient is not showing any signs or symptoms, treatment for multiple myeloma isn’t always necessary. These signs include bone pain, especially in the chest or spine; nausea; constipation; appetite loss; mental confusion; frequent infections; fatigue; weight loss; weakness or numbness in the legs; and excessive thirst, medical treatment is needed to help control the disease.
Because cancer cells don’t mature and then die like normal cells do, they accumulate, eventually overwhelming the production of healthy cells. In the bone marrow, myeloma cells crowd out healthy white blood cells and red blood cells, leading to fatigue and an inability to fight infections.
The myeloma cells continue trying to produce antibodies like healthy plasma cells, but the cancerous cells produce abnormal antibodies that the body can’t use. Instead, the abnormal antibodies (monoclonal proteins, or M proteins) build up in the body and cause damage to the kidneys and the bones (even raising the risk of broken bones). It can also lead to various type of organ failure and death.
Deciphering the “blueprint” of cancer cells – outlining how cancer cells hijack specific pathways for uncontrolled proliferation – will lead to more efficient ways to fight it.
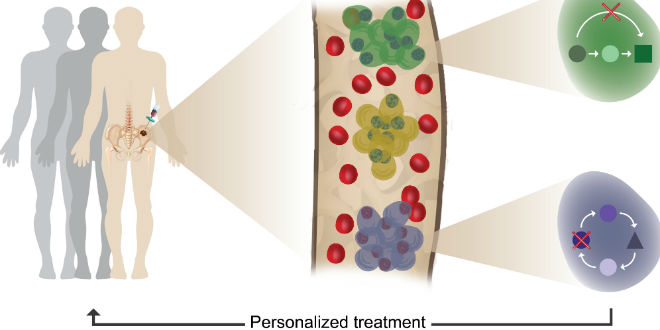
A combined effort by scientists from the Weizmann Institute of Science in Rehovot, Israel, and physicians from major hemato-oncology departments in this country succeeded in creating detailed profiles of myeloma cancer in both pre-cancer stages – in newly diagnosed multiple myeloma patients as well as after treatment and relapse. These detailed blueprints will help in future precision diagnosis and treatment of this disease.
Multiple myeloma is the second-most common type of blood cancer. Despite many years of research and a significant recent improvement in the survival of myeloma patients with new immunotherapy treatments, many patients do not do well; all relapse.
A major stumbling block, in diagnosing myeloma disease is the fact that each patient is unique and current blood tests are incapable of identifying early disease onset and classifying which patient should receive which treatment. For example, patients whose routine blood tests reveal some hallmarks of the disease in an early and pre-cancerous stage are followed closely with a “watch and wait” strategy. However, every year, 1% of them will lose in this “Russian roulette” and develop the full-blown myeloma disease.
Until recently, there had been no way to discriminate between those that will develop the full-blown disease and those that will not.
Dr. Guy Ledergor, a physician who also earned a doctorate, and Dr. Assaf Weiner in Prof. Ido Amit’s group from the Weizmann Institute’s immunology department, thought that a highly sensitive method developed by the group known as single cell RNA sequencing may provide a new approach to understanding the makeup of multiple myeloma and develop new and more effective approaches to diagnose and treat this devastating disease.
Their study and the sensitive method that has been used for the first time in the world was just published in the prestigious journal Nature Medicine. It is hoped that it will help dvance Israel in the forefront of myeloma patient diagnosis and treatment.
Working together with Prof. Amos Tanay of the biological regulation and computer science and applied mathematics departments and Prof. Gabi Barbash of the institute’s Bench to Bedside Program, the team carried out a nationwide effort to recruit all of the hemato-oncology departments in Israel.
The new method sequences the RNA in thousands of individual cells from the patient blood or bone marrow, making it possible to capture the specific gene program that is active in each individual cell. To understand the myeloma cancer blueprint, the scientists first generated a high-resolution model of normal plasma cells by sequencing tens of thousands of cells from healthy individuals undergoing hip-replacements who served as a control group.
The plasma cells of the control patients were very similar within and across individuals, basically showing a single, common blueprint of normal plasma cells. Comparing the normal blueprint to the blueprints of patients showed that the blueprint of myeloma cancer cells is extremely heterogeneous, with every patient showing its own, unique blueprint and with some patients demonstrating several tumor clones displaying more than a single blueprint in one patient.
The team showed that it’s possible to identify even a very small number of malignant cells in the blood at very early (pre-cancer) stage. This allows for much more precise diagnosis and informed choices about treatments for each patient based on his “personalized” disease. As a result, it will also be possible to follow and monitor these patients with significantly less painful blood tests, replacing today’s painful bone marrow biopsies.
Eventually the method might help multiple myeloma patients receive earlier and more precise treatments, and possibly prevent the relapse that often follows chemotherapy.
Barbash noted that their findings are just the beginning for applying single cell RNA-seq technologies to clinical-genomic research. “We are now developing similar collaborations between hospital-based researchers and basic-research scientists to advance diagnosis and treatment for other illnesses.”
Single-cell genomic analysis until now was confined to a small number of research labs, he continued. “We are constantly pushing the boundaries of the technology in ways that will make it a major clinical discovery and diagnostics tool,” added Amit. “Such tracking may be useful for diagnosing early signs of many malignancies in the precancerous stage or post-chemotherapy relapse, thus becoming part of a personalized approach. It will enable decisions to be made based on the individual profiles of each patient’s cells at every stage of disease. These profiles are already used to create targeted therapies against the cancer cells.”
The researchers used a machine-learning approach for automatically identifying malignant cells out of tens of thousands of cells. “We are entering an era in which measuring big-data and using machine learning will provide clinicians new insights and understanding into devastating diseases like multiple myeloma.” said Weiner. “In the future, physicians will hopefully be able to track the disease in real time and treat each patient according to a personalized disease profile, possibly even before symptoms appear,” concluded Ledergor.